18 November 2020
Download Presentation Slide Here
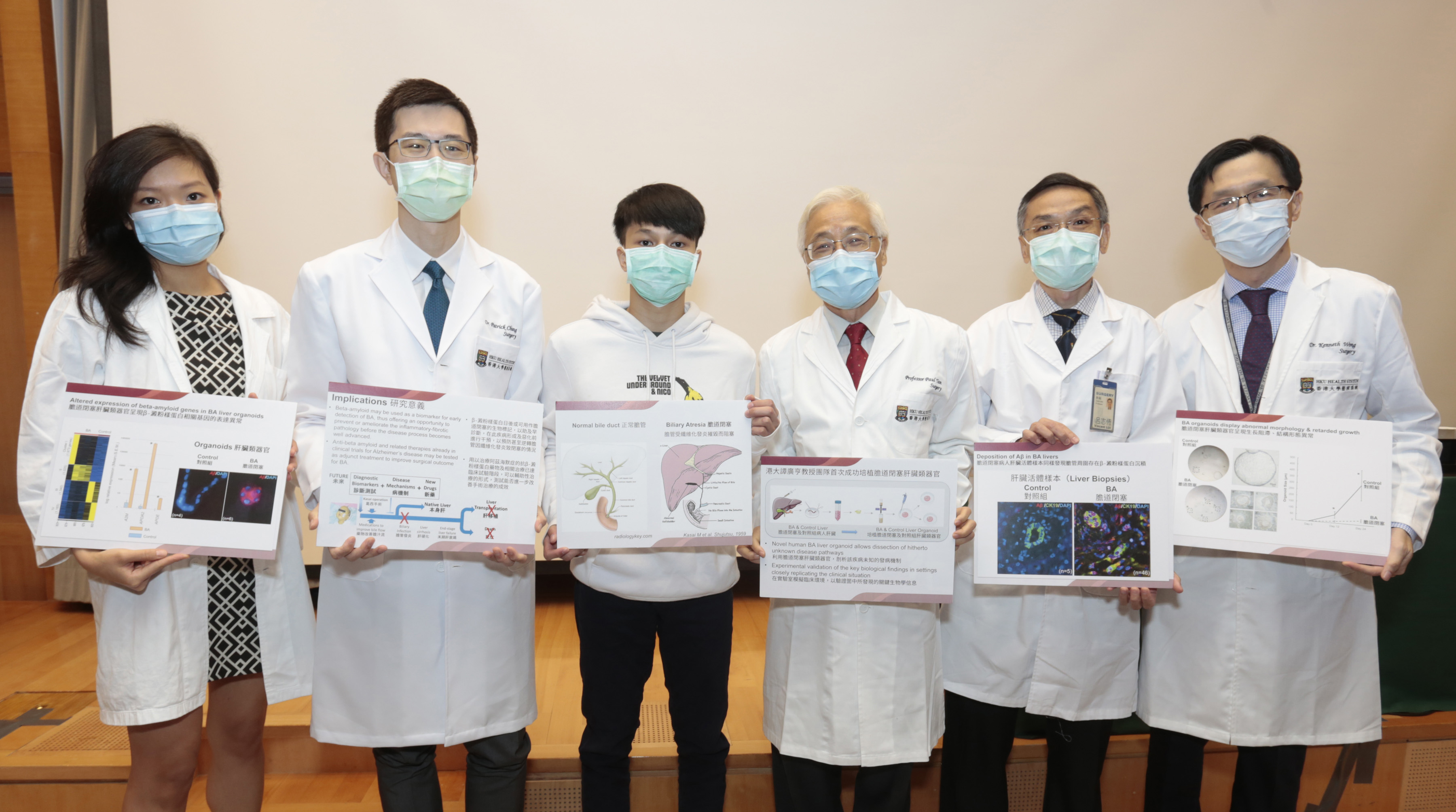
A research team at LKS Faculty of Medicine, The University of Hong Kong (HKUMed) in collaboration with overseas and Mainland researchers discovered abnormal beta-amyloid deposition to be a novel disease mechanism for biliary atresia (BA). The ground-breaking finding provides insights into new diagnostic and therapeutic strategies for BA, a devastating newborn liver disease with unknown cause till now. The novel findings have been published in the leading academic journal, Journal of Hepatology. [link to the publication]
Background
Biliary atresia (BA) is a life-threatening congenital anomaly characterised by progressive fibro-inflammatory obstruction of the entire biliary tract in newborn babies, resulting in liver failure if untreated. BA is especially common among Asians, with an incidence of 1.2 to 2.0 per 10,000 live births in Hong Kong and Mainland China respectively. The only treatment is surgery but the results are unsatisfactory. Kasai portoenterostomy (removal of diseased biliary tract outside the liver, followed by the joining of the bowel to the residual diseased biliary tract within the liver) restores bile drainage in only 60-70% of patients. Even for these survivors, most will face life-long complications including bile infection, liver failure and portal hypertension. The root cause for the failure to improve treatment outcome is the lack of understanding of the underlying disease mechanisms for BA due primarily to the absence of a valid disease model for BA.
An organoid is a cell/tissue-derived culture system that can mimic a real-life (in vivo) organ “in a dish”. Human liver organoids have been described but until now, none has been successfully developed for BA. HKUMed’s novel human BA liver organoid allows dissection of hitherto unknown disease pathways and experimental validation of the key biological findings in settings closely replicating the clinical situation.
Research findings
Herein, the team successfully grew liver organoids from BA patients and controls, and found that BA organoids showed retarded growth, abnormal morphology (structure and form), and disturbed cell polarity (apex-base orientation) and organisation. Analysis of gene-readouts (transcriptome) of BA organoids revealed a developmental shift from bile duct cells to liver cells and, most importantly, altered beta-amyloid-related gene expression. Beta-amyloid accumulation around the bile ducts was confirmed in liver biopsies of BA patients. Finally, administration of beta-amyloid to control organoids induced the BA-specific morphology. In brief, HKUMed research team is the first to use liver organoids in the study of BA and identify beta-amyloid deposition, which is also the main pathological feature of Alzheimer’s disease and Cerebral Amyloid Angiopathy, around BA bile ducts. The finding reveals a novel disease mechanism for BA, paving the way for innovative diagnostic and therapeutic strategies to radically improve treatment outcome.
“Our study provides the first evidence that beta-amyloid deposition is a key factor in the cause and deterioration of biliary atresia which till now is an incurable disease. Instead of empirical surgical treatment when the disease is well-advanced, a novel beta-amyloid-based strategy may allow firstly, early diagnosis for prevention/early treatment; secondly, identification of poor risk patients for more aggressive treatment; and thirdly, improvement in treatment outcome by the use of anti-beta amyloid drugs. Our unique ‘patient-based’ liver organoid bridges the gap between laboratory experimentation and clinical application and allows new diagnostic and therapeutic paradigms to be tested safely and accurately in a patient-specific manner,” said Professor Paul Tam Kwong-hang, Li Shu-Pui Professor of Surgery, Director of Dr Li Dak-Sum Research Centre and Chair Professor of Paediatric Surgery, Department of Surgery, HKUMed, who led the research.
Significance of the study
Biliary atresia (BA) is a major healthcare and socio-economic burden with little progress in its treatment in the past 50 years.
The findings of this study have significant implications in precision medicine. Firstly, the identification of beta-amyloid deposition in BA suggests that it may be used as a biomarker for early detection of BA, thus offering an opportunity to prevent or ameliorate the inflammatory-fibrotic pathology before the disease process becomes well advanced. Secondly, anti-beta amyloid and related therapies already in clinical trials for Alzheimer’s disease may be tested as adjunct treatment to improve surgical outcome for BA. Thirdly, HKUMed’s unique ‘patient-based’ BA organoid faithfully replicates patient-specific pathobiology. As BA is believed to be a heterogeneous disease, patient-derived liver organoid testing may help to differentiate good and poor responders to novel therapeutics, advancing precision medicine for BA and related diseases.
About the research team
The research was led by HKUMed Professor Paul Tam Kwong-hang, Li Shu-Pui Professor of Surgery, Director of Dr Li Dak-Sum Research Centre and Chair Professor of Paediatric Surgery, Department of Surgery, HKUMed. Dr Rosana Ottakandathil Babu, former post-doctoral fellow of Professor Tam’s team, Dr Vincent Lui Chi-hang, Associate Professor, and Dr Yan Chen, Honorary Assistant Professor, Department of Surgery, HKUMed are co-first authors. Professor Urban Lendahl, Department of Cell and Molecular Biology, Karolinska Institutet, Stockholm, Sweden and Professor Bin Wang, Department of General Surgery, Shenzhen Children’s Hospital, Shenzhen, Mainland China are co-corresponding authors. Other researchers include Dr Rachel Yiu Sze-wan, MBBS-PhD graduate, The University of Hong Kong; Dr Yongqin Ye, Department of General Surgery, Shenzhen Children’s Hospital, Shenzhen, Mainland China; Dr Ben Niu and Professor Michael Qi Zhang, Department of Biological Sciences, Center for Systems Biology, The University of Texas at Dallas, Texas, USA; Dr Zhongluan Wu, Research Assistant, Dr Michelle Yu On-na, Resident Specialist, Dr Patrick Chung Ho-yu, Clinical Assistant Professor, and Dr Kenneth Wong Kak-yuen, Clinical Associate Professor, Chief of Division of Paediatric Surgery, Department of Surgery, HKUMed; and Dr Ruizhong Zhang and Professor Huimin Xia, Department of Pediatric Surgery, Guangzhou Women and Children’s Medical Center, Guangzhou, Mainland China.
Department of Cell and Molecular Biology, Karolinska Institutet, Stockholm, Sweden; The University of Texas at Dallas, Texas, USA; Shenzhen Children’s Hospital, Shenzhen, Mainland China; and Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou, Mainland China also contributed to the research
This work was supported in part by the Health and Medical Research Fund (03143476) and University-Industry Collaboration Programme, Innovation and Technology Fund, Innovation and Technology Commission (UIM/300), HKSAR Government; HKU Dr Li Dak-Sum Research Centre matching grant; and Sanming Project of Medicine in Shenzhen, China (SZSM201812055); and National Natural Science Foundation of China (81771629).
Media enquiries
Please contact LKS Faculty of Medicine of The University of Hong Kong by email (medmedia@hku.hk).


Follow HKUMed