05 January 2023
Download presentation diagrams
A research team led by Professor Shen Jiangang from the School of Chinese Medicine, LKS Faculty of Medicine, The University of Hong Kong (HKUMed) reveals that Angong Niuhuang Wan (AGNHW), a classical Chinese medicinal formula, could extend therapeutic window for ischemic stroke by 30 minutes and reduce hemorrhagic transformation and mortality in ischemic stroke with Thrombolytic therapy. The findings have been recently published in the scientific journal Chinese Medicine [link to the publication].
Background
Stroke is one of the leading causes of disability and death in human diseases worldwide. In Hong Kong, nearly three thousand people die of stroke each year, it is the fourth fatal disease at present.1 About 85% stroke patients suffer from ischemic stroke2 when the blood supply to part of the brain is interrupted or reduced, preventing brain tissue from getting oxygen and nutrients. Time is brain and for every minute that stroke is not treated, it has been estimated that up to 1.9 million brain neurons may be lost.
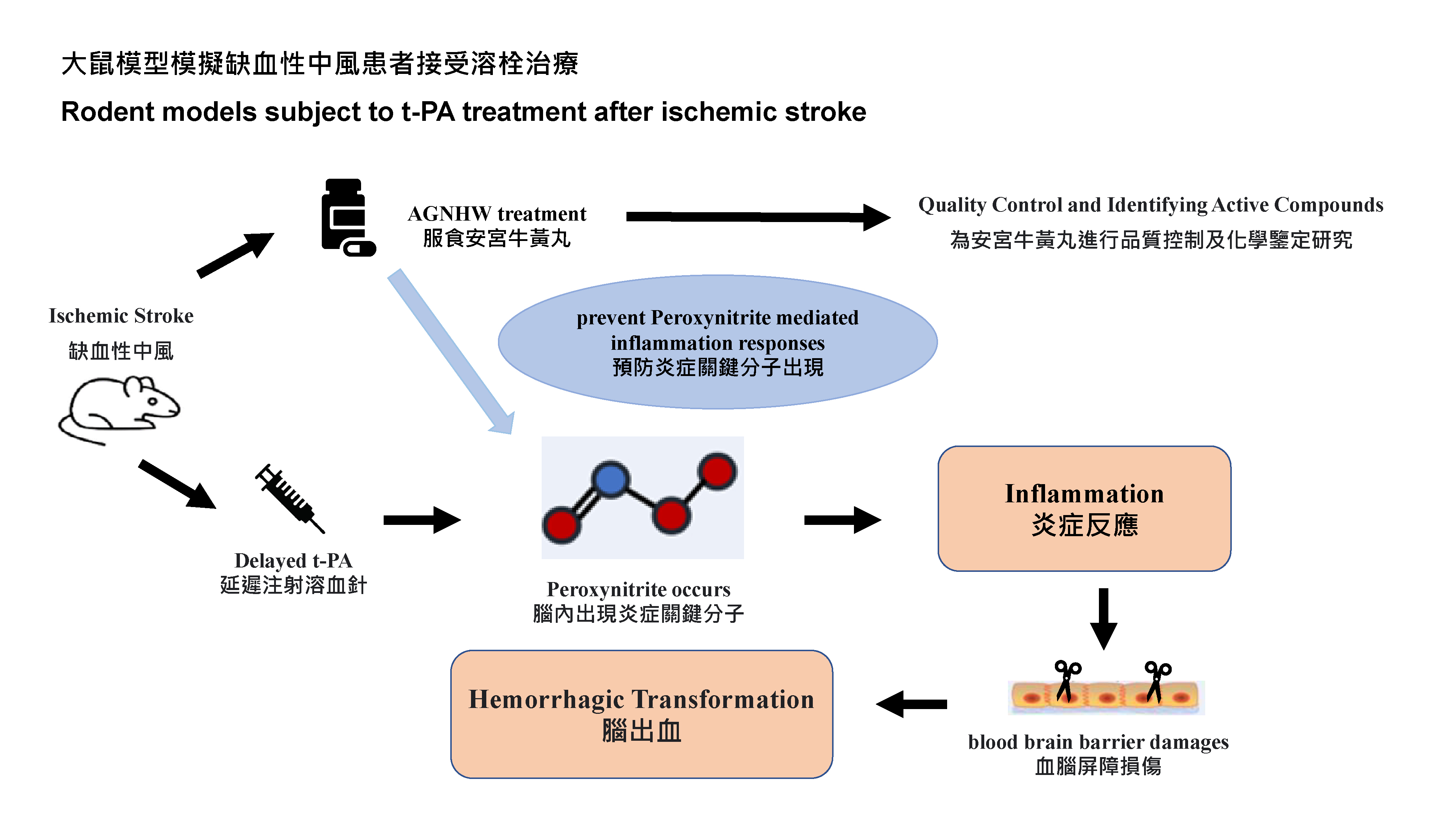
To significantly lower mortality rates and presence of complications, patients must be treated by the thrombolytic therapy3 (t-PA)4 within four and a half hours of onset. Otherwise it would increase the risk of developing intracerebral heamorrhage instead, contributing to the increased mortality. Blood-brain barrier-disruption5 is a key event that worsens the stroke outcome. Earlier treatment for stroke is associated with better outcome, especially within the 4.5 hours therapeutic window. However, less than 10% of ischemic stroke patients could benefit from t-PA. Focusing on the intracerebral heamorrhage aroused by receiving the delayed t-PA treatement, the research team has previously reported that a key molecule related oxidative stress and inflammation named peroxynitrite6 was found based on their previous animals and clinical experiments. Scavenging the molecule could protect the blood-brain barrier integrity; the team also revealed that AGNHW contains Baicalin that it could help with the scavenge.
Research methods and findings
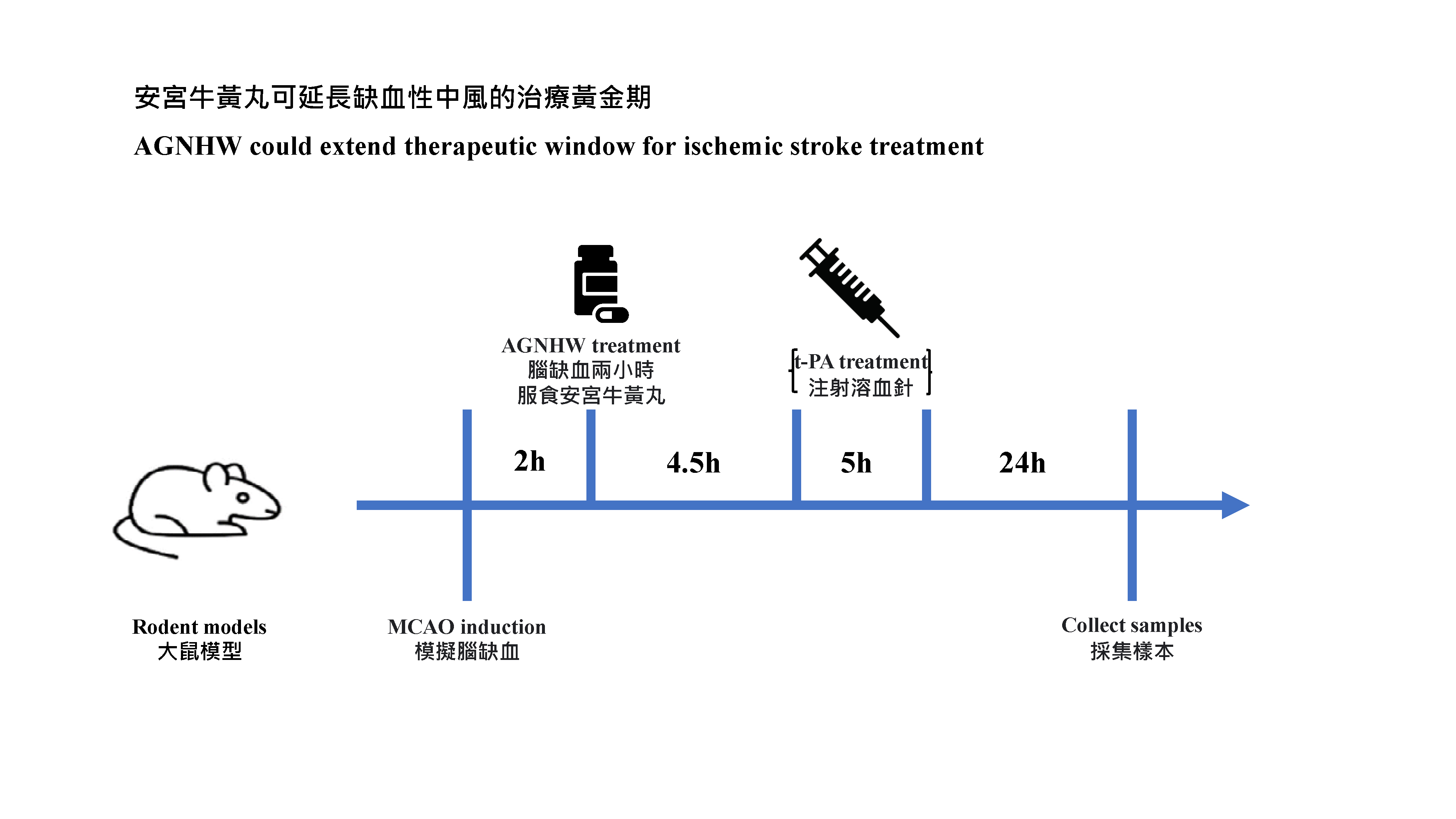
Researchers first performed quality control study and chemical identification of AGNHW. In animal experiments, rodent models were subjected to 5 hours of middle cerebral artery occlusion. AGNHW (257 mg/kg) was given orally at 2 hours post cerebral ischemia in the animal model (The dosage of AGNHW used for the animals was equivalent to the regular dosage of one capsule per day). In the previous studies, the research team has confirmed that the dosage used equivalent to normal adult dosage (one capsule per day for one week) was safe, with no liver and kidney toxicity, and no excessive heavy metal residues were observed. The team measured hemorrhagic transformation levels in ischemic brains and assessed the severity of the blood-brain barrier damage. They used the modified neurologic severity score test to assess neurological functions. In addition, the researchers investigated the neuroprotective mechanisms of AGNHW and the contained active compounds by using brain microvascular endothelial cells cultured in vitro, and found that six active compounds7 in AGNHW can remove the key cytotoxic molecules that cause inflammation and the blood brain barrier damages.
The research team has proved that AGNHW significantly ameliorated the blood-brain barrier damage and brain edema, reduced hemorrhagic transformation, enhanced neurological function, and decreased the mortality rates in transient cerebral ischemia rat models with delayed t‑PA treatement at 5 hours after ischemia stroke, i.e., the therapeutic window has been extended by 30 minutes. The neuroprotective mechanisms of AGNHW treatment were found to be attributed to prevent peroxynitrite mediated inflammation responses and related molecular events that led to the blood-brain barrier damage, brain edema, reduced hemorrhagic transformation in ischemic brains with delayed t‑PA treatment.
These results have suggested that AGNHW may serve as an adjuvant therapy with t-PA to protect blood-brain barrier integrity, reduce hemorrhagic transformation, and improve the treatment outcome of ischemic stroke. AGNHW could be a first-aid treatment at early stage of ischemic stroke and potentially reduce the side effects of t-PA.
Significance of the study
t-PA has a limited golden treatment time window of 4.5 hours after the onset of ischemic stroke, and many patients cannot arrive at the hospital for medical imaging diagnosis and treatment within such a narrow time window. Unlike other prescription drugs, AGNHW has become a first-aid medicine for emergency situations in Chinese communities.
‘Our research team previously reported that the short-term use of AGNHW in regular doses was safe and effective in the treatment of ischemic stroke. A recent meta-analysis also showed that AGNHW can improve neurological function in patients with acute cerebral infarction and acute cerebral hemorrhage. The research will enter the clinical trial stage soon as researchers are encouraged by the data obtained from animal tests. Further clinical trial studies may lead to a new therapeutic strategy to rescue ischemic stroke patients and extend the golden treatment time window of t-PA by using AGNHW as emergency treatment. Since AGNHW is a traditional Chinese medicine formula with multiple ingredients, a Chinese medicine practitioner should be consulted first before taking.’ remarked by Professor Shen Jiangang from the School of Chinese Medicine, HKUMed, who led the study.
About the research team
The research was led by Professor Shen Jiangang, principal investigator and Professor of the School of Chinese Medicine, HKUMed. Dr Chen Hansen and Dr Tsoi Bun, former postdoctoral researchers and Dr Luo Yunxia, former research exchange student, School of Chinese Medicine, HKUMed. Other collaborators include Professor Qi Suhua and Professor Gu Bing from Xuzhou Medical University, China.
Acknowledgements
This work was supported by Hong Kong General Research Fund (GRF No. 17118717, 17105220), Areas of Excellence Scheme, Research Grant Council, Hong Kong SAR, China (AoE/P-705/16) and Shenzhen Basic Research Plan Project (SIRI/04/04/2015/06). The study was also supported by Contract Research from Beijing Tong Ren Tang Chinese Medicine Company Limited as a component of the funding support from Innovation and Technology Fund, Hong Kong SAR, China (UICP No. UIM/289).
- www21.ha.org.hk/smartpatient/SPW/en-us/Disease-Information/Disease/?guid=29ac1219-3d68-4378-a2bd-09e111da3650
- Stroke is fundamentally divided into ischemic stroke and hemorrhagic stroke.
- By injecting the thrombolytic agent , tissue plasminogen activator (t-PA), could dissolve the thrombus and restore the blood flow to the brain, reducing damage to the brain cells and thereby disability and death caused by stroke.
- Tissue plasminogen activator (t-PA) remains the only thrombolytic drug approved by the U.S. Food and Drug Administration (FDA) for treating acute ischemic stroke.
- The blood-brain barrier is a highly selective semipermeable membrane that restricts the entrance of the blood-borne substances into the brain thus maintaining cerebral homeostasis to allow proper function of the neurons. Blood-brain barrier-disruption after ischemic stroke is a key event that precedes various neurological disorders while the contributing factors that govern the process remain unclear.
- Peroxynitrite: This is a representative active nitrogen species produced by ischemia-reperfusion injury caused by vascular recanalisation after cerebral ischemia. It can cause inflammation and enhance MMP-9 activity, thereby aggravating blood-brain barrier damage and brain injury.
- Six active compounds: epiberberine, coptisine, baicalin, palmatine, berberine, and baicalein.
Media enquiries
Please contact LKS Faculty of Medicine of The University of Hong Kong by email (medmedia@hku.hk).

Follow HKUMed