10 August 2020
A study led by a research team of LKS Faculty of Medicine, The University of Hong Kong (HKUMed) in collaboration with Harvard Medical School, provided the first population-based evidence that a commonly used anti-osteoporosis drug, bisphosphonate, was associated with reduced risk of pneumonia and pneumonia mortality in patients with hip fracture, compared to non-treatment and other anti-osteoporosis treatment. The findings are now published in the latest issue of the Journal of Bone and Mineral Research, one of the highest-ranked journals in the field [link to the publication].
Hip fracture and pneumonia events
Hip fracture is a major health problem in older persons. According to a latest study1, it is projected that the number of cases will be over 27,000 in 2050 and the direct cost of hip fracture will increase from HK$600 million dollars in 2018 to HK$1.9 billion dollars in 2050. Hip fracture leads to substantial morbidity and mortality, with one-tenth to one-third patients dying in the first year following a hip fracture. Pneumonia is one of the major causes of death in hip fracture. Thus, there is a clinical concern about preventing or reducing the risk of these life-threatening outcomes.
Drugs for hip fracture
Nitrogen-containing bisphosphonates (N-BPs; such as alendronate and zoledronate) are commonly used in treating osteoporosis and preventing fracture. However, the drug is under-used worldwide, at least in part due to patients’ concerns about its serious but rare side effects. In fact, N-BPs may have extra-skeletal effects, which might be beneficial for the health of other tissues.
Animal studies showed that the highest concentration of N-BPs was detected in respiratory tract among other non-bone tissues after oral ingestion or intravenous injection in animal. In a previous post-hoc analysis of randomised controlled trial (RCT) of zoledronate, zoledronate was associated with reduced risk of pneumonia mortality. Thus, it is possible that N-BPs may reduce risk of pneumonia. However, the relationship between N-BPs and pneumonia is less studied. It is well recognised that RCTs usually have limited number of participants who are rarely representative of the actual patient population who will be taking these drugs. A large observational study using real data is valuable to extend the findings from RCTs.
Research method
The research team conducted a population-based study using an anonymised territory-wide healthcare database of the Hong Kong Hospital Authority. The aim was to investigate the risk of pneumonia with and without use of N-BPs in patients with hip fracture.
The study included over 50,000 patients with newly diagnosed hip fracture from 2005 through 2015. The team examined the treatment in these patients and matched the N-BP-treated and non-N-BP-treated patients by propensity scores (a statistical matching technique) to ensure their similarity. The matched patients were followed until 31 December 2016, and their risks of pneumonia and pneumonia-mortality were compared.
Research findings
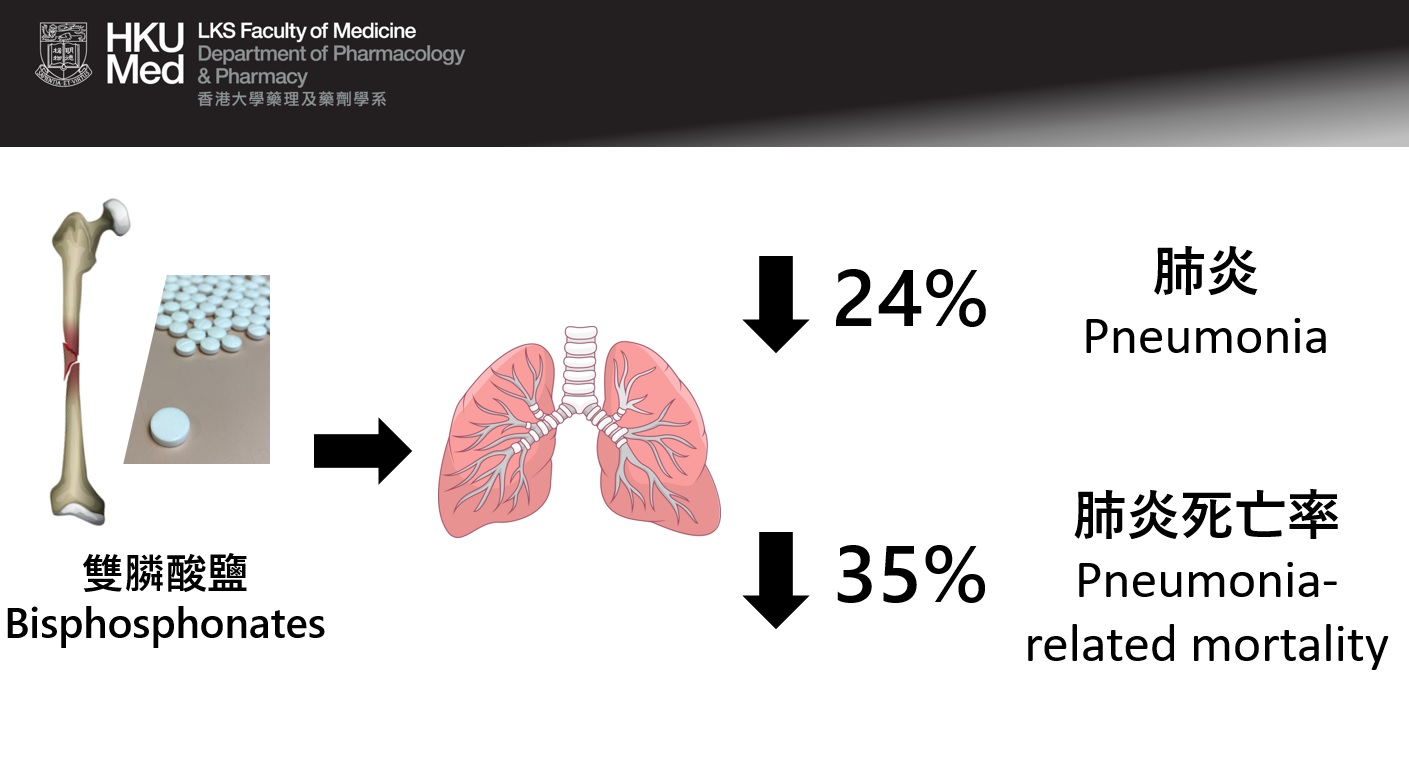
There were 4,041 N-BP-treated patients and 11,802 non-treated patients in the final analysis. In the first year of follow-up, the team found that 6.9 per 100 persons vs. 9 per 100 persons of pneumonia being observed in N-BP-treated and non-treated patients, respectively, leading to 24% reduction in risk of pneumonia. N-BP-treated patients were also associated with 35% reduction in risk of pneumonia mortality, compared to non-treated patients. This beneficial tendency was observed when comparing N-BP-treated patients with patients treated with other anti-osteoporosis medications.
Recommendation of the study
This is the first population-based study examining the risk of pneumonia events with and without N-BP treatment in patients with hip fracture. Patients are commonly under-treated with osteoporosis drugs after hip fracture, even though clinical guidelines recommend treatment. This study highlights the importance of N-BP treatment after hip fracture, encouraging its use in clinical practice.
The work was supported by Seed Fund for Basic Research (No. 201811159065) from The University of Hong Kong.
About the research team
This study was led by Dr Cheung Ching-lung, Assistant Professor of Department of Pharmacology and Pharmacy, HKUMed, in collaboration with Professor Douglas P Kiel, Professor of Medicine, Harvard Medical School, Director of the Musculoskeletal Research Center, and Senior Scientist for the Institute for Aging Research at Hebrew SeniorLife, US; Professor Richard B Hubbard, Professor, Division of Epidemiology and Public Health, School of Medicine, University of Nottingham, Nottingham, UK; and Dr Wallis Lau Cheuk-yin, Lecturer, Research Department of Practice and Policy, University College London (UCL) School of Pharmacy, London, UK. Other researchers included Professor Ian Wong Chi-kei, Lo Shiu Kwan Kan Po Ling Professorship in Pharmacy, Professor and Head of Department of Pharmacology and Pharmacy, HKUMed; Professor Annie Kung Wai-chee, Honorary Clinical Professor of Department of Medicine, HKUMed; Dr Sing Chor-wing and Dr Gloria Li Hoi Yee, Post-doctoral fellow of Department of Pharmacology and Pharmacy, HKUMed.
- Cheung CL, Ang SB, Chadha M, Chow ES, Chung YS, Hew FL, Jaisamrarn U, Ng H, Takeuchi Y, Wu CH, Xia W, Yu J, Fujiwara S. An updated hip fracture projection in Asia: The Asian Federation of Osteoporosis Societies study. Osteoporos Sarcopenia. 2018 Mar;4(1):16-21.
Media enquiries
Please contact LKS Faculty of Medicine of The University of Hong Kong by email (medmedia@hku.hk).



Follow HKUMed